APIs & Intermediates Custom Development
BOC Sciences focuses on R&D and production services for APIs & Intermediates. We have accumulated more than 20 years of project experience, focusing on custom R&D, process development, production and sales of APIs & Intermediates, developed a rich R&D service line, and formed a unique core competitiveness. With the increasing number of service lines, we are still making industrial layouts and striving to expand into new fields. At present, the company's APIs & Intermediates are divided into two major segments, namely featured APIs and patented original research drugs.
Active Pharmaceutical Ingredient
Active pharmaceutical ingredients (APIs) are the biologically active components of a drug that produce the desired therapeutic effect. APIs are the basic chemical compounds that make a drug effective in treating a specific disease or condition. These substances are responsible for the pharmacological activity of the drug and are formulated along with inactive ingredients to form the final dosage form such as tablets, capsules, injections, or liquids. APIs can be obtained from a variety of sources including plants, animals, microorganisms, and synthetic chemicals. Depending on the nature of the drug and its intended use, APIs can have different physical and chemical properties. In drug development, the selection of the right API is crucial as it determines the efficacy, safety, and stability of the final drug product. In terms of therapeutics, APIs can be classified into different categories based on their intended use such as antibiotics, anti-inflammatory drugs, antiviral drugs, cardiovascular drugs, central nervous system (CNS) drugs, and oncology drugs.
Pharmaceutical Intermediates
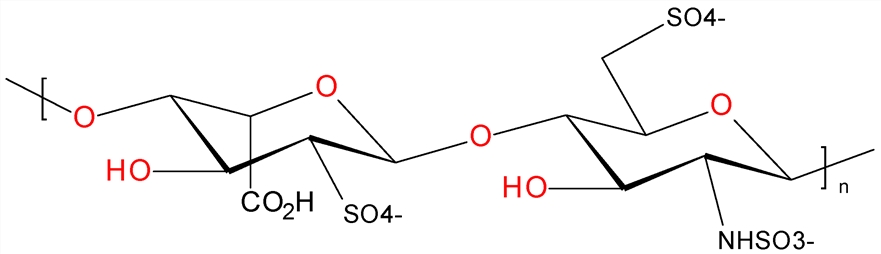
Drug intermediates are chemical compounds produced during the synthesis of APIs. These intermediate compounds undergo further chemical reactions to ultimately form active pharmaceutical ingredients. Drug intermediates are essential in the manufacturing process of APIs as they serve as building blocks or precursor molecules for the synthesis of complex drug compounds. The production of drug intermediates involves various chemical reactions and purification processes to ensure the quality and purity of the final API. Drug intermediates can be classified based on their specific chemical structure, function in the synthetic pathway, or role in the final drug formulation. These intermediate compounds play a key role in optimizing the efficiency and yield of API synthesis. Drug intermediates are classified based on their role in the synthetic pathway, including key intermediates, side chain precursors, ring closure reagents, protecting groups, and coupling agents.
CDMO Manufacturing
BOC Sciences is a leading global chemical supplier and contract research organization that provides comprehensive API & Intermediate custom development services. Our experienced chemists and scientists work closely with clients to design and synthesize API & Intermediates to their specifications, ensuring the highest purity, quality and regulatory compliance. Our API & Intermediate development services cover a wide range of activities, including route exploration, process optimization, impurity profiling and scale-up.
CRO Services
As a large chemical service organization, BOC Sciences has reached R&D cooperation projects with hundreds of pharmaceutical companies and biotechnology companies. We provide one-stop service from target verification, to chemical synthesis, pharmacological efficacy testing, pharmacokinetics and early toxicological testing. Our eight technology platforms, including continuous flow chemistry, enzyme catalysis, and crystallization technology, can provide extensive and effective professional chemical services, providing a strong technical guarantee for customers to screen drug candidates. We also have first-class experimental equipment and provide professional analysis and development services.
CDMO Services
BOC Sciences' CDMO service can provide further process optimization and scale-up services for drug development. Our services include design, screening and development of new production routes; evaluation and optimization of key process parameters based on the concept of "Quality by Design (QbD)"; research and establishment of quality standards; scale-up of kilo-scale laboratories and pilot plants production and cGMP production. The company has four large pharmaceutical cGMP production bases. With dozens of advanced production lines and internationally recognized quality management systems in place.
What Can We Offer?
BOC Sciences' expertise in chemical synthesis enables us to efficiently manufacture APIs and intermediates that are important for the treatment and management of a wide range of diseases. We have the ability to design compounds that target specific cancer cells or signaling pathways, helping to develop more effective and less toxic treatment options for patients. Our commitment to innovation and quality assurance ensures that the compounds we develop meet stringent standards.
| Alzheimer's disease | Inflammation | Immune system |
| Parkinson's disease | Neurological disease | Cardiovascular/blood system |
| Cancer | Diabetic | Dermatology |
| Cancer immunotherapy | Hypertensive | Digestive system |
| Infection | Obesity | Endocrine/metabolic system |
R&D Labs

Our R&D center is equipped with advanced experimental and analytical instruments, such as LC-MS/MS, HPLC, prep-chromatography, GC/MS, IC and controlled-release dosage research equipment, which can meet most of the pharmaceutical chemical synthesis services. More than 65% of the employees in the R&D team have a master's degree or above. We specialize in deeply integrating a broad knowledge of organic chemistry, peptide chemistry, analytical chemistry, structural chemistry, and more into your projects. At the same time, our efficient project management methods and good quality management system also help us to better complete customer projects. BOC Sciences has taken years of endeavor to build our high- standard quality control and assurance system. In addition, BOC Sciences has a 600-square-meter Class C pilot plant and a 200-square-meter Class D pilot plant, as well as a 100-square-meter Class C QC testing plant that meets cGMP requirements. The 80-square-meter Class C plant has P2 laboratory qualifications and has an aseptic filling and preparation production line.
Production Facilities

BOC Sciences passed the ISO9001 quality system certification in 2020. In combination with cGMP, GLP and other regulatory requirements, it has established a quality management system that meets the requirements of drug research and pilot production, including electronic systems such as material management, equipment and facility management, and personnel management. Our main production facility is located in China. We have dozens of cGMP compliant production lines equipped with more than 100 reactors ranging from 20 L to 2,000 L for gram to ton scale reactions. In addition to the large-scale production of intermediates, we can also provide the production of registered starting materials. We are committed to applying a Quality by Design (QbD) methodology to every batch of our products.
Why Choose BOC Sciences?
Custom synthesis expertise: BOC Sciences has a team of experienced chemists and scientists who specialize in custom synthesis of APIs and intermediates.
High quality standards: Quality is a top priority for BOC Sciences, and the company adheres to strict quality control measures throughout the API and intermediate development process.
Materials controllable and traceable: From raw material sourcing to final product testing, BOC Sciences ensures that every step meets the highest quality standards and regulatory requirements.
Scalability and flexibility: Whether small-scale synthesis is required for research purposes or large-scale production is required for commercialization, we can adapt to different project sizes and timelines.
Comprehensive analytical capabilities: Equipped with advanced analytical instrumentation including spectroscopic techniques, chromatography and other methods to ensure the purity, identity and stability of synthesized compounds.
Regulatory compliance: Committed to meeting regulatory standards and guidelines for API and intermediate development to ensure the safety and efficacy of the final drug product.
Collaborative approach: Working closely with customers throughout the API and intermediate development process to promote open communication and collaboration.
Timely delivery: Our streamlined processes and project management ensure that customers receive API and intermediate products in a timely manner.
In addition to API & Intermediate development, BOC Sciences also provides API manufacturing services to clients seeking reliable and cost-effective solutions for chemical synthesis projects. Whether clients require custom API synthesis or intermediate development support, we are a trusted partner. If you are interested in our APIs & Intermediates custom development service, please contact us immediately.